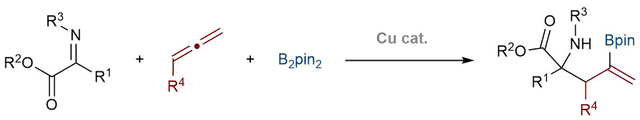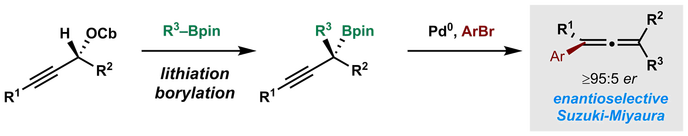our Publications. |
#32 |
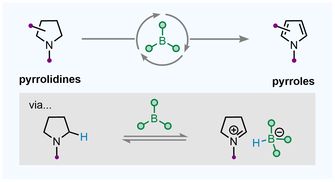
A. Alvarez-Montoya, J. P. Gillions, L. Winfrey, R. R. Hawker, K. Singh, F. Ortu, Y. Fu, Y. Li,* A. P. Pulis* B(C6F5)3-Catalyzed Dehydrogenation of Pyrrolidines to Form Pyrroles ACS Catalysis 2024, DOI: 10.1021/acscatal.3c05444 |
#31 |
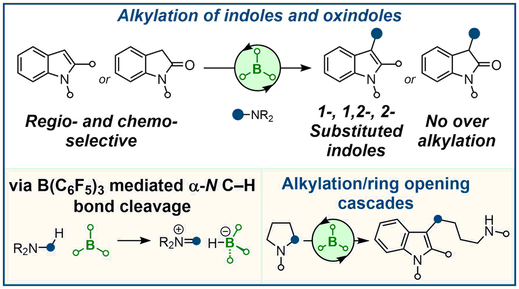
S. A. Elsherbeni, R. L. Melen*, Alexander P. Pulis*, Louis C. Morrill* Accessing Highly Substituted Indoles via B(C6F5)3-Catalyzed Secondary Alkyl Group Transfer J. Org. Chem. 2024, DOI: 10.1021/acs.joc.4c00025 |
#30 |
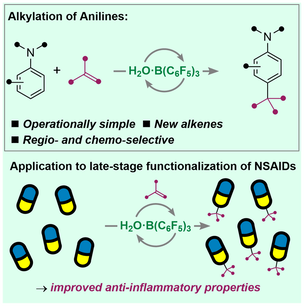
L. Winfrey, L. Yun, G. Passeri, K. Suntharalingam,* A. P. Pulis* H2O·B(C6F5)3-Catalyzed para-Alkylation of Anilines with Alkenes Applied to Late-Stage Functionalization of Non-Steroidal Anti-Inflammatory Drugs Chem. Eur. J. 2024, e202303130 |
#29 |
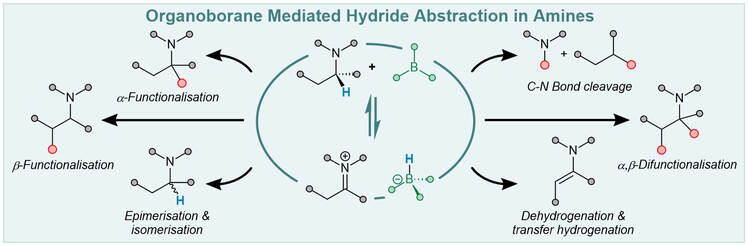
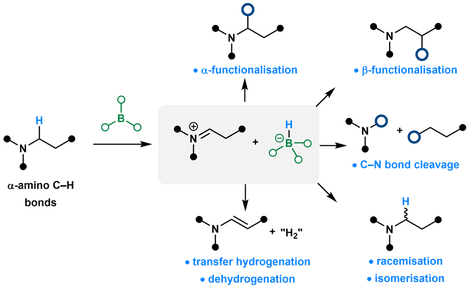
J. P Gillions, S. A Elsherbeni, L. Winfrey, L. Yun, R. L. Melen,* L. C. Morrill,* A. P. Pulis* Recent Advances in Catalysis using Organoborane Mediated Hydride Abstraction Synlett 2023, DOI:10.1055/a-2111-9629 Invited contribution to celebrate 60 years of Donald Matterson’s boron homologation Selected for the cover graphic by the Editorial Board |
#28 |
S. Basak, L. Winfrey, B. A. Kustiana,R. L. Melen,* L. C. Morrill,* A. P. Pulis* Electron deficient borane-mediated hydride abstraction in amines: stoichiometric and catalytic processes Chem. Soc. Rev. 2021, 50, 3720-3737 |
#27 |
S. Basak, A. Alvarez-Montoya, L. Winfrey, R. L. Melen,* L. C. Morrill,* A. P. Pulis* B(C6F5)3‑Catalyzed Direct C3 Alkylation of Indoles and Oxindoles ACS Catal. 2020, 10, 4835-4840 |
#26 |
A. P Pulis,* L. Winfrey, A. Alvarez-Montoya Uncovering the Synthetic Potential of Frustrated Lewis Pairs in Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, 2019, p 1. Invited Review |
#25 |
J. Yan, A. P. Pulis, G. J. P. Perry, D. J. Procter Metal‐Free Synthesis of Benzothiophenes by Twofold C−H Functionalization: Direct Access to Materials‐Oriented Heteroaromatics Angew. Chem. Int. Ed. 2019, 58, 15675 |
#24 |
T. Jia, M. J. Smith, A. P. Pulis, G. J. P. Perry, D J. Procter Enantioselective and Regioselective Copper-Catalyzed Borocyanation of 1-Aryl-1,3-Butadienes ACS Catalysis 2019, 9, 6744 Highlighted in SYNFACTS 2019, 15, 1012 |
#23 |
Z. He, A. P. Pulis, G. J. P. Perry, D. J. Procter Pummerer chemistry of benzothiophene S-oxides: Metal-free alkylation and arylation of benzothiophenes Phosphorus, Sulfur, and Silicon and the Related Elements 2019, 194, 669 |
#22 |
Z. He, A. P. Pulis, D. J. Procter* The interrupted Pummerer reaction in a sulfoxide‐catalyzed oxidative coupling of 2‐naphthols Angew. Chem. Int. Ed. 2019, 58, 7813 Top 10% most downloaded ACIE article during the year after publication |
#21 |
M. Šiaučiulis, N. Ahlsten, A. P. Pulis, D. J. Procter* Transition metal‐free cross‐coupling of benzothiophenes and styrenes in a stereoselective synthesis of substituted (E,Z)‐1,3‐dienes Angew. Chem. Int. Ed. 2019, 58, 8779 |
#20 |
K. Yeung, F. J. T. Talbot, G. P. Howell, A. P. Pulis, D. J. Procter* Copper-catalyzed borylative multi-component synthesis of quaternary α-amino esters ACS Catalysis 2019, 9, 1655 Highlighted in SYNFACTS 2019, 15, 571 |
#19 |
K. Yang, A. P. Pulis , G. J. P. Perry, D. J. Procter* Transition-metal-free synthesis of C3-Aarylated Benzofurans from Benzothiophenes and Phenols Org. Lett. 2018, 20, 7498 |
#18 |
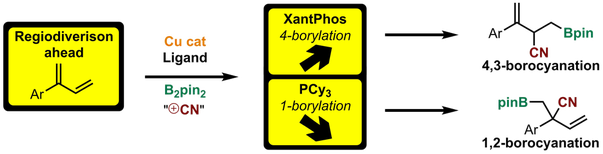
T. Jia, Q. He, R. E. Ruscoe, A. P. Pulis, D. J. Procter* Regiodivergent copper catalyzed borocyanation of 1,3-dienes Angew. Chem. Int. Ed. 2018, 57, 11305 |
#17 |
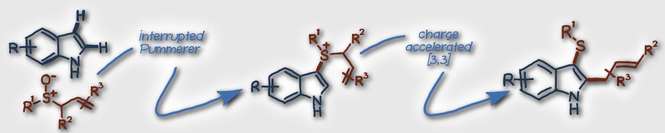
Miles H. Aukland, Fabien J. T. Talbot, José A. Fernández-Salas, Matthew Ball, Alexander P. Pulis, and David J. Procter* An Interrupted Pummerer/Nickel‐Catalysed Cross‐Coupling Sequence Angew. Chem. Int. Ed. 2018, 57, 9785 |
#16 |
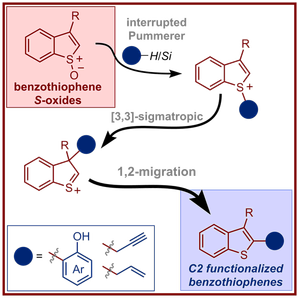
Z. He, H. J. Shrives, J. A. Fernández-Salas, A. Abengózar, J. Neufeld, A. P. Pulis and D. J. Procter* Synthesis of C2 Substituted Benzothiophenes via an Interrupted Pummerer/[3,3]-Sigmatropic/1,2-Migration Cascade of Benzothiophene S-Oxides Angew. Chem. Int. Ed. 2018, 57, 5759 |
#15 |
M. Šiaučiulis, S. Sapmaz, A. P. Pulis and D. J. Procter* Dual vicinal functionalisation of heterocycles via an interrupted Pummerer coupling/[3,3]-sigmatropic rearrangement cascade Chem. Sci. 2018, 9, 754 Highlighted in SYNFACTS 2018, 14, 368 |
#14 |
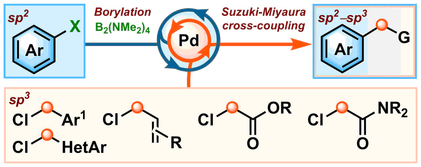
L. Whitaker, H. Y. Harb* and A. P. Pulis* One-pot borylation/Suzuki-Miyaura sp2–sp3 cross-coupling Chem. Commun. 2017, 53, 9364 |
#13 |
A. P. Pulis, A. Varela, C. Citti, P. Songara, D. Leonori and V. K. Aggarwal* Asymmetric Synthesis of Tertiary Alcohols and Thiols via Non-Stabilized Tertiary α-Oxy- and α-Thio-Organolithiums Angew. Chem. Int. Ed. 2017, 56, 10835 Hot Paper; Highlighted in SYNFACTS 2017, 13, 1193 |
#12 |
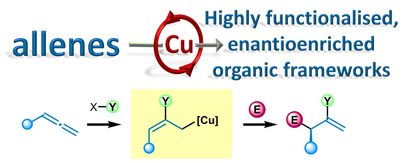
A. P. Pulis*, K. Yeung and D. J. Procter* Enantioselective copper catalysed, direct functionalisation of allenes via allyl copper intermediates Chem. Sci. 2017, 8, 5240 Review |
#11 |
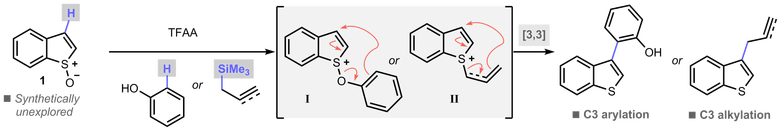
H. J. Shrives, J. A. Fernández-Salas, C. Hedtke, A. P. Pulis and David J. Procter* Regioselective synthesis of C3 alkylated and arylated benzothiophenes Nature Commun. 2017, 8, 14801 |
#10 |
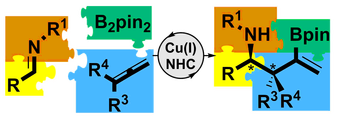
K. Yeung, R. E Ruscoe, J. Rae, A. P. Pulis and D. J. Procter* Enantioselective Generation of Adjacent Stereocenters in a Copper-Catalyzed Three-Component Coupling of Imines, Allenes, and Diboranes Angew. Chem. Int. Ed. 2016, 55, 11912 Hot Paper; Highlighted in SYNFACTS 2016, 12, 1267 |
#09 |
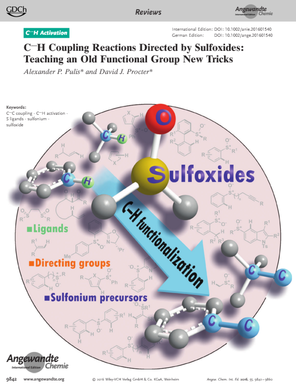
A. P. Pulis* and D. J. Procter* C−H Coupling Reactions Directed by Sulfoxides: Teaching an Old Functional Group New Tricks Angew. Chem. Int. Ed. 2016, 55, 9842 Review |
#08 |
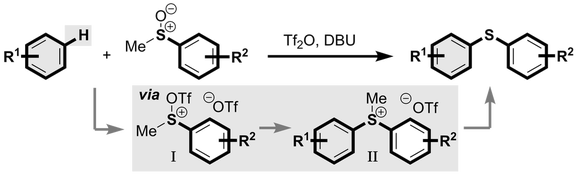
J. A. Fernández-Salas, A. P. Pulis and D. J. Procter* Metal-free C–H thioarylation of arenes using sulfoxides: a direct, general diaryl sulfide synthesis Chem. Commun. 2016, 52, 12364 |
#07 |
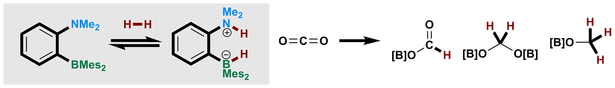
É. Rochette, M. -A. Courtemanche, A. P. Pulis, W. Bi and F.-G. Fontaine* Ambiphilic Frustrated Lewis Pair Exhibiting High Robustness and Reversible Water Activation: Towards the Metal Free Hydrogenation of Carbon Dioxide Molecules 2015, 20, 11902 Invited for a Special Issue on "The Reactivity of Frustrated Lewis Pairs" |
#06 |
M.-A. Courtemanche, A. P. Pulis, É. Rochette, M.-A. Légaré, D. W. Stephan* and F.-G. Fontaine* Intramolecular B/N frustrated Lewis pairs and the hydrogenation of carbon dioxide Chem. Commun. 2015, 51, 9797 |
#05 |
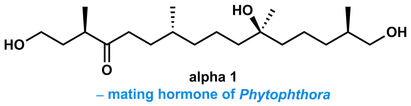
A. P. Pulis, P. Fackler and V. K. Aggarwal* Short Stereoselective Synthesis of the Phytophthora Universal Mating Hormone Alpha 1 using Lithiation/Borylation Reactions. Angew. Chem. Int. Ed. 2014, 53, 4382 |
#04 |
S. Roesner, C. A. Brown, M. Mohiti, A. P. Pulis, R. Rasappan, D. J. Blair, S. Essafi, D. Leonori and V. K. Aggarwal* Stereospecific conversion of alcohols into pinacol boronic esters using lithiation–borylation methodology with pinacolborane Chem. Commun. 2014, 50, 4053 Highlighted in SYNFACTS 2014, 10, 631 |
#03 |
A. P. Pulis, D. J. Blair, E. Torres and V. K. Aggarwal* Synthesis of Enantioenriched Tertiary Boronic Esters by the Lithiation/Borylation of Secondary Alkyl Benzoates J. Am. Chem. Soc. 2013, 135, 16054 Highlighted in SYNFACTS 2014, 10, 81; Organic Chemistry Portal; and Methods in Organic Synthesis |
#02 |
B. M. Partridge, L. Chausset-Boissarie, M. Burns, A. P. Pulis, and V. K. Aggarwal* Enantioselective Synthesis and Cross-Coupling of Tertiary Propargylic Boronic Esters Using Lithiation-Borylation of Propargylic Carbamates Angew. Chem. Int. Ed. 2012, 51, 11795 Highlighted in SYNFACTS 2013, 9, 198; Methods in Organic Synthesis |
#01 |
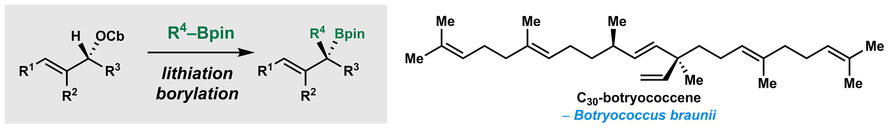
A. P. Pulis and V. K. Aggarwal Synthesis of Enantioenriched Tertiary Boronic Esters from Secondary Allylic Carbamates. Application to the Synthesis of C30 Botryococcene J. Am. Chem. Soc. 2012, 134, 7570 |